Last updated March 8, 2018 at 9:47 am
As mammals go, bats should be completely screwed up and dysfunctional. The fact that they aren’t, and have evolved to produce around 1,200 species throws up some particularly curly questions for scientists.

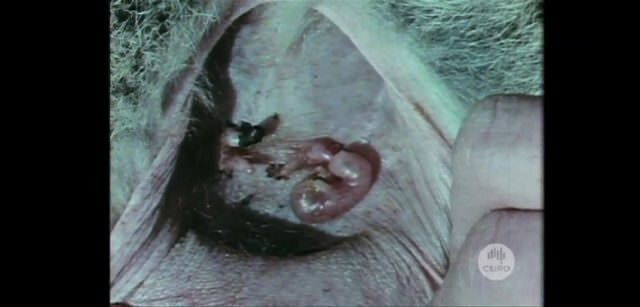
The flying fox or fruit bat is renowned as a carrier of Australian bat lyssavirus belong to a group of viruses that include rabies. Credit: iStock
There are three reasons why bats shouldn’t be as successful as they are.
First off, they fly – which is weird for a mammal, but, more importantly, means that their cells are put under enormous stress and often leak fragments of DNA from their nuclei into the surrounding cell cytoplasm. This is generally regarded by biologists as not a healthy thing.
Second, they live comparatively long lives, which means that the results of the damage they incur become ever more serious as time rolls on.
And third, they tend to have an awful lot of virus DNA sloshing around inside them, because they are superbly adapted to functioning as reservoirs for a wide range of diseases – think Ebola, Marburg, Hendra, Nipah and SARS, for instance – which kill humans but leave their hosts untouched.
Just how bats manage to stay healthy when the evidence suggests that the lot of them should have long ago dropped dead from a combination of cell damage and a boiling mass of multiple infections has long exercised the curiosity of biologists, geneticists and virologists alike.
What is the bats’ secret?
New research has succeeded in uncovering at least part of the reason for the animals’ improbable success.
A team led by Peng Zhou of the Chinese Academy of Sciences, in Wuhan, China, has established that bats share genetic adaptations that result in an antiviral pathway that is radically different to the equivalent found in other mammals. The research is published in the journal Cell Host & Microbe.
In most mammals the presence of DNA, or RNA, in cell cytoplasm sends an alarm signal to the immune system, which activates proteins called interferon type 1 (IFN) that stimulate white blood cells to attack, and also catalyse inflammation. In moderation, this is a good thing, but overstimulation of IFN response has been linked to the onset of severe autoimmune diseases.
Bats don’t suffer from any such problems, which is strange because at first blush – awash as their cells are with broken bits of their own DNA, bacterial DNA and viral RNA – they should have IFN levels off the scale.
Research into why they don’t has been widespread. In 2016, a team led by Lin-Fa Wang of Duke-National University in Singapore revealed that bats are missing an entire family of genes – known as PYHIN – that in other mammals function as sensors for the presence of foreign (or damaged) DNA, and stimulate both interferon production and inflammation.
Interferon regulation
Other studies have found that genes regulating innate immunity, and those governing interferon regulation, are significantly different to those found in mice and humans.
Peng and his colleagues set out to discover whether abnormal DNA floating about in cell cytoplasm – that is, either DNA from infective agents or broken bits from the bat itself – influence how the animal’s immune system functions.
They concentrated on an immune sensor system known as STING (for “STimulator of INterferon Genes”) and found that a key downstream regulator that catalyses system response, known as S358, was entirely missing. The absence of the regulator – present in all other mammals – the researchers write, “strongly suggests a weakened bat STING in the context of IFN activation”.
In other words, bat immune systems are always switched on, but run at very low levels.
Peng says this is probably an evolutionary adaption to the rigours of flight. Given the amount of oxidative damage to its own DNA that occurs while airborne, a robust and strong immune system would simply trigger crippling inflammation and autoimmune distress.
“Adaptation to flight likely caused positive selection of multiple bat innate immune and DNA damage repair genes,” he says.
A side effect of the changes to immune system sensitivities is that bats became exquisitely set up to act as reservoirs for a wide variety of pathogens. Their systems are strong enough to prevent resident virus or bacterial populations making them sick, but not so strong that they vanquish the invaders altogether.
“In human history, we have been chasing infectious diseases one after another,” says Zhou, “but bats appear to be a ‘super-mammal’ to these deadly viruses”.
Education Resource:
Bats’ Evolutionary Trick
Follow us on Facebook, Twitter and Instagram to get all the latest science.