Last updated January 31, 2018 at 4:59 pm
Macrophages could provide one answer to counter the rise of the superbugs.
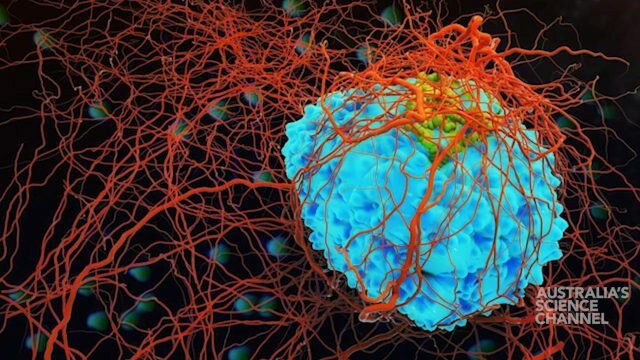
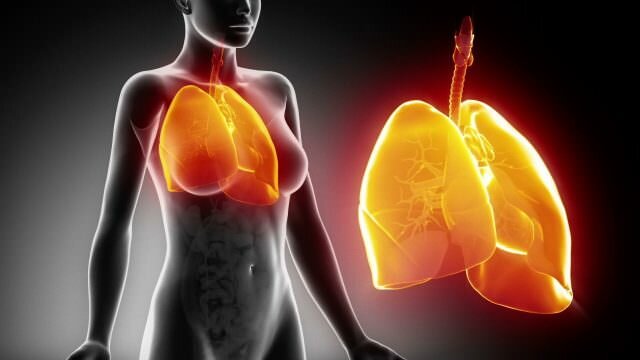
An artist’s impression of a macrophage destroying bacteria. Credit 7activestudio/istock
From Blade Runner to The Walking Dead, we’re constantly seeing glimpses of a dystopian future that could become a reality if the next few centuries go terribly wrong. And while hyper-realistic worker androids and zombies are pure fiction (for now), there’s another kind of nightmare that’s worryingly close.
Right now, at least 700,000 people die every year from drug-resistant infections worldwide, and that number is predicted to rise to 10 million a year by 2050. Why? Because our antibiotics are failing, and we’re not making new ones fast enough to stop harmful ‘superbugs’ from becoming immune.
If we can’t find a solution to the rise of superbugs, we could revert to the life our ancestors led, where dental surgery can be fatal; you risk losing your leg because of a simple scratch while gardening; and as for elective surgery, it’s just too risky to contemplate.
While substances with antibiotic properties have been used for centuries, they were discovered by chance, and until recently, weren’t all that effective – particularly for infections inside the body. It wasn’t until the end of the 19th century that scientists started to gain an understanding of how antimicrobials actually worked.
In 1928, Alexander Fleming identified penicillin as a molecule that kills or slows the growth of certain kinds of bacteria, but it took another decade for methods of industrial-scale production and purification to be developed. By 1945, it was put into widespread use as an antibiotic.
Fast-forward to now, and thanks to the overuse of antibiotics in both the pharmaceutical and agricultural industries, we run the risk of turning back the clock with the rise of a new breed of superbugs – bacteria that have evolved so fast, they can no longer be killed by many, most, or even any, antibiotics.
“Fortunately, cases where people are dying because there is no effective therapy left to treat them with are still rare,” Dr Mark Blaskovich, a senior research chemist at the Institute for Molecular Bioscience (IMB) at The University of Queensland in Australia, said.
“But certainly around the world there are increasing numbers of people dying, because their infection can’t be treated by any available antibiotic.”
Tuberculosis is a case in point. In the 19th century, it was estimated to be the cause of nearly 15 per cent of all deaths, but by the end of the 20th century, it had all but disappeared from developed countries. Now that trend has been reversed, with an estimated 500,000 new cases of multidrug-resistant tuberculosis (MDR-TB) being reported worldwide each year.
And everyone has heard the horror stories of a hospital stay turned deadly thanks to Methicillin-resistant Staphylococcus aureus (MRSA), where the normally benign bacterium Staphylococcus aureus enters the bloodstream during surgery.
“Hospitals are the breeding ground for the most serious infections, but antibiotic resistance is increasingly being acquired in the community. Once they are out in the environment, people can spread them around – and global travel is assisting that,” Dr Blaskovich said.
Scientists at the IMB are at the forefront of the fight to stay one step ahead of the superbugs.
Dr Blaskovich and his team are working on ways to develop more potent versions of current antibiotics based on peptides, or by finding older antibiotics that were discovered decades ago but not used. These older antibiotics are promising because they’ve not been overused, so bacteria are less likely to have built up resistance to them.
Although new antibiotics are being developed, bacteria can rapidly evolve to defend against them
But IMB researchers are also pursuing other angles. Professor Matt Sweet, Director of IMB’s Centre for Inflammation & Disease Research, is targeting the immune system as an alternate approach to fighting infections.
“Our work is trying to understand how it detects and responds to infections,” Professor Sweet said.
“Although new antibiotics are being developed, bacteria can rapidly evolve to defend against them. Of course, such approaches to combat bacterial infections are still incredibly important, because if there’s no antibiotic that can be used, you’re in serious trouble. But we’re trying another approach – to manipulate or ‘train’ the immune system to better defend against infection.”
Professor Sweet’s research focuses on characterising genes and pathways in immune cells called macrophages, which both drive inflammation and are involved in the clearance of bacterial pathogens.
“Macrophage” literally means big eater.
“These are cells that are present in every tissue in our body. They detect danger – if we cut ourselves or have an infection, for example,” Professor Sweet said. “These cells are often long-lived, and they use a number of different strategies to defend against pathogens.”
Sweet describes macrophages as the ‘garbage trucks of the body’, gobbling up and destroying pathogens and unwanted debris: “Once they take up the pathogen, they engage a range of different pathways to try to kill the ingested microorganism.”
The process by which a macrophage engulfs a target microbe was first described in the late 1800s
But this is not always effective. Pathogens actively evade the immune system – and many actually do so by hiding out inside the macrophages.
“It’s a cunning strategy if you’re a pathogen. A number of different viruses, bacteria, parasites, and fungi can all do this,” Professor Sweet said.
Although phagocytosis – the process by which a macrophage engulfs a target microbe – was first described in the late 1800s, our knowledge of the strategies used by macrophages to kill the ingested pathogen is still incomplete. Over the past decade, scientists have discovered several antimicrobial strategies that are used by macrophages to successfully overcome infections.
“In recent times, we found that macrophages use toxic concentrations of metal ions to kill intracellular bacteria,” Professor Sweet said.
Metals such as zinc and iron are essential for survival. Many of the enzymes in both our own cells and in bacteria need zinc and iron to function. One of the responses of the immune system involves starving bacteria of these metals. But another strategy relies on the delivery of toxic concentrations of zinc and copper to kill the bacteria in macrophages.
“What we found a number of years ago, in collaboration with other researchers at The University of Queensland, is that macrophages take up copper from the extracellular space to clear bacterial pathogens within these cells,” Professor Sweet said.
“More recently, we and others have found that zinc is used in a similar way – macrophages and other immune cells subject intracellular bacteria to a zinc stress response.”
Professor Sweet’s lab is focussed on the idea that if they can understand how macrophages defend against bacteria, they might be able to reprogram these cells to be better equipped to fight superbugs.
In recent times, there’s been a lot of interest in this approach, which Sweet says reflects the concern about the rapid increase in multidrug-resistant bacteria. And that’s pushed the field into thinking about different ways of tackling antibiotic resistance.
“We do have some exciting new data in development,” he says. “These findings suggest that we can indeed manipulate macrophages to more effectively clear intracellular bacterial infections.”
This article was first published on the website of The University of Queensland’s Institute for Molecular Bioscience. It is reprinted here with permission.